In this laboratory, we use our knowledge and experience in the area of protein structure and function to determine the chemical mechanisms employed by interesting biological factors. The major focus of the laboratory is in understanding regulation in the transcription factor NF-κB (pronounced, "En Ef Kappa Bee") signal transduction pathway. NF-κB is a relatively small class of proteins that respond to diverse stimuli by activating the expression of numerous genes. NF-κB responsive genes include many of the key components of the cellular survival program including inflammatory cytokines, mediators and effectors of both innate and adaptive immunity, and inhibitors of apoptosis. Although proper NF-κB function is integral to a cell's ability to fight off infection and respond to stress, too much of an NF-κB response can contribute to states of chronic inflammation such as arthritis, asthma, multiple sclerosis, and colitis. Recently, it has been shown that chronically inflamed tissues can serve as hotbeds for tumor formation. Cellular processes that recognize and kill tumors in healthy tissues fail to function effectively under the influence of the NF-κB cell survival program. Chronic inflammation due to hyperactive NF-κB has also been shown to contribute to sclerotic formation in arteries and heart disease.
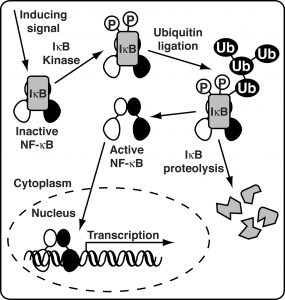
The prototypical NF-κB functions as a heterodimer of p50 and p65 (aka RelA) subunits. NF-κB is present in the cytoplasm of all cells as an inactive factor in complex with a member of the IκB ("Eye Kappa Bee") inhibitor protein family. Diverse NF-κB-inducing stimuli lead to activation of the IκB kinase complex (IKK). IKK is a large multisubunit complex that specifically phosphorylates a pair of serine amino acid side chains in the amino-terminal region of NF-κB complex-associated IκB. Once phosphorylated, IκB is recognized by a specific E3 Ubiquitin-protein ligase complex leading to its poly-ubiquitinylation. The 26 S Proteasome can then recognize and proteolyze IκB. Removal of IκB renders NF-κB active. It rapidly translocates from the cytoplasm to the nucleus where it binds specifically to DNA elements within the promoter regions of target genes and activates their transcription (figure to the right).
We are currently working on the following two NF-κB-related projects:
1) IKK structure and function. IKK is a multisubunit kinase complex that specifically phosphorylates IκB. Purification of IKK from cytokine-induced HeLa cells revealed that it is composed of three subunits. These are referred to as IKKα (IKK1), IKKβ (IKK2), and IKKγ (NEMO, FIP3). Although IKKα and IKKβ are highly conserved protein subunits, they differ significantly in their cellular function. For example, the IKKβ subunit has been shown to be responsible for activating NF-κB in response to inflammatory stimuli by catalyzing the attachment of two phosphates near the amino-terminus of the classical IκB proteins. Furthermore, IKKβ itself is subject to phosphorylation-dependent regulation of its own catalytic activity. We are interested in understanding the detailed mechanisms of substrate specificity and phosphorylation-dependent regulation of the IKKβ subunit.
2) Nuclear IκB structure and function. The classical NF-κB inhibitor proteins, IκBα, IκBβ, and IκBε, function primarily in the cell cytoplasm by masking NF-κB nuclear localization signals and blocking DNA binding. However, two additional classes of IκB proteins are also integral to NF-κB regulation.
The proteins p105 and p100 play dual roles as IκB proteins and precursors of the mature NF-κB p50 and p52 subunits, respectively. The identification of a third general class of IκB proteins that function exclusively in the nucleus has been made recently. The nuclear IκB proteins include Bcl-3, IκBζ (MAIL, INAP), and IκBNS. These proteins all show similar properties: their expression is regulated by NF-κB; they rapidly accumulate in the nucleus; they have modulatory effects on NF-κB-dependent expression of specific target genes. We have shown that in contrast to classical IκB proteins, the nuclear IκBζ protein binds preferentially to the NF-κB p50 homodimer. We also found that formation of this protein-protein complex does not remove the NF-κB homodimer from binding to target DNA. We are currently interested in studying how assembly of an IκBζ:NF-κB p50:DNA complex in the nucleus activates the expression of specific NF-κB responsive genes such as the cytokine interleukin-6 (IL-6).
Other Projects in which we are currently involved include:
3) Structure and function of a myosin motor protein co-chaperone. In collaboration with the Drosophila muscle laboratory of Dr. Sanford I. Bernstein in the Department of Biology at San Diego State University, we are studying the structure and function of the factor UNC-45 that works in muscles to repair the misfolded heads of myosin motor proteins.
4) Antibodies that target signaling lipids. The hydrolytic products of membrane phospholipids are potent signaling molecules. In collaboration with local biotechnology companies, we are studying the structures of antibodies that have been raised to recognize specific biologically active antigens. This lead to our discovery of an antibody that uses calcium as a bridging factor in antigen recognition and binding of its lipid antigen, sphingosine-1-phosphate (S1P). We are currently interested in characterizing the metal binding properties of this antibody and understanding whether the use of calcium in antigen binding is a common mechanism in mammalian immunity.

